Stainless Steel Passivation: What It Is Used For
Introduction
Passivation is a fundamental chemical treatment for stainless steel, designed to enhance its corrosion resistance properties. This process involves applying an acid solution that removes surface contaminants and promotes the formation of a chromium oxide layer on the metal’s surface. This protective layer is essential to ensure that stainless steel maintains its anti-corrosive characteristics in aggressive environments. Passivation is widely used in industries such as food, pharmaceutical, chemical, and marine applications, where durability and structural integrity are crucial.
What Is Stainless Steel Passivation?
Stainless steel passivation is a chemical process that improves the material’s corrosion resistance by eliminating surface impurities and promoting the formation of a stable passive layer. This layer, composed mainly of chromium oxide, is highly resistant to chemical attacks and protects the underlying metal from corrosion.
Mechanism of Passivation
The chromium present in stainless steel reacts with oxygen during passivation, forming a thin chromium oxide layer on the surface. This layer is invisible to the naked eye but is extremely adherent and self-healing. If the metal surface is damaged, the passive layer quickly reforms in the presence of oxygen, ensuring continuous protection against corrosion.
Stages of the Passivation Process
Preliminary Cleaning
Description: Before passivation, the stainless steel surface must be cleaned to remove oils, greases, and other contaminants that could interfere with the process. This cleaning can be carried out using specific detergents, degreasers, or alkaline solutions.
Advantages:
- Ensures a clean and uniform surface, essential for effective passive layer formation.
Acid Solution Treatment
Description: Stainless steel is immersed or sprayed with an acid solution, commonly nitric or citric acid, which removes metallic impurities such as free iron particles and promotes the formation of the passive layer.
Critical Parameters:
- Acid Concentration: Varies depending on the type of steel and final application.
- Immersion Time: Typically from 15 minutes to several hours, depending on the surface condition.
- Temperature: Can be increased to accelerate the process but must be controlled to avoid surface damage.
Rinsing and Drying
Description: After acid treatment, the metal is thoroughly rinsed to remove any acidic residues. This rinsing is essential to prevent corrosion beneath the passive layer.
Techniques Used: Rinsing with deionized water, air drying, or hot-air drying to avoid water spots and surface oxidation.
Quality Control
Description: The passivated surface is inspected to ensure the passive layer has formed correctly and no residual contamination remains. Corrosion resistance tests can be conducted to verify the effectiveness of passivation.
Control Tools: Immersion tests, salt spray tests, measurement of passive layer thickness.
Table: Comparison Between Nitric Acid and Citric Acid Passivation Treatments
| Characteristic | Nitric Acid Passivation | Citric Acid Passivation |
|---|---|---|
| Composition | Nitric acid (20-50%) | Citric acid (4-10%) |
| Effect | Removal of heavy metallic contaminants, formation of a dense passive layer | Gentle removal of contaminants, environmentally friendly |
| Environmental Impact | Requires careful waste management | Less impactful, biodegradable |
| Applications | Heavy industries, high-strength stainless steel | Food, pharmaceutical, biotechnological sectors |
| Advantages | High effectiveness on contaminated steels | Environmental and health safety |
Applications of Passivation
Food and Pharmaceutical Industry
Description: In these industries, stainless steel is used for building equipment, tanks, piping, and work surfaces that must remain sterile and corrosion-resistant. Passivation ensures these surfaces remain free from contamination and protected from corrosion.
Advantages:
- Improves corrosion resistance in humid and acidic environments.
- Essential to prevent contamination of food and pharmaceutical products.
Chemical and Petrochemical Industry
Description: Stainless steel components used in aggressive chemical environments, such as reactors, heat exchangers, and piping, require superior corrosion protection. Passivation improves durability and reduces maintenance frequency.
Advantages:
- Increases component lifespan.
- Reduces maintenance costs and plant downtime.
Marine Applications
Description: Stainless steel exposed to marine environments undergoes high chemical stress due to the presence of chlorides and humidity. Passivation helps prevent corrosion and pitting, protecting the structural integrity of components.
Advantages:
- Long-lasting protection against corrosion and rust.
- Improves resistance to localized corrosion.
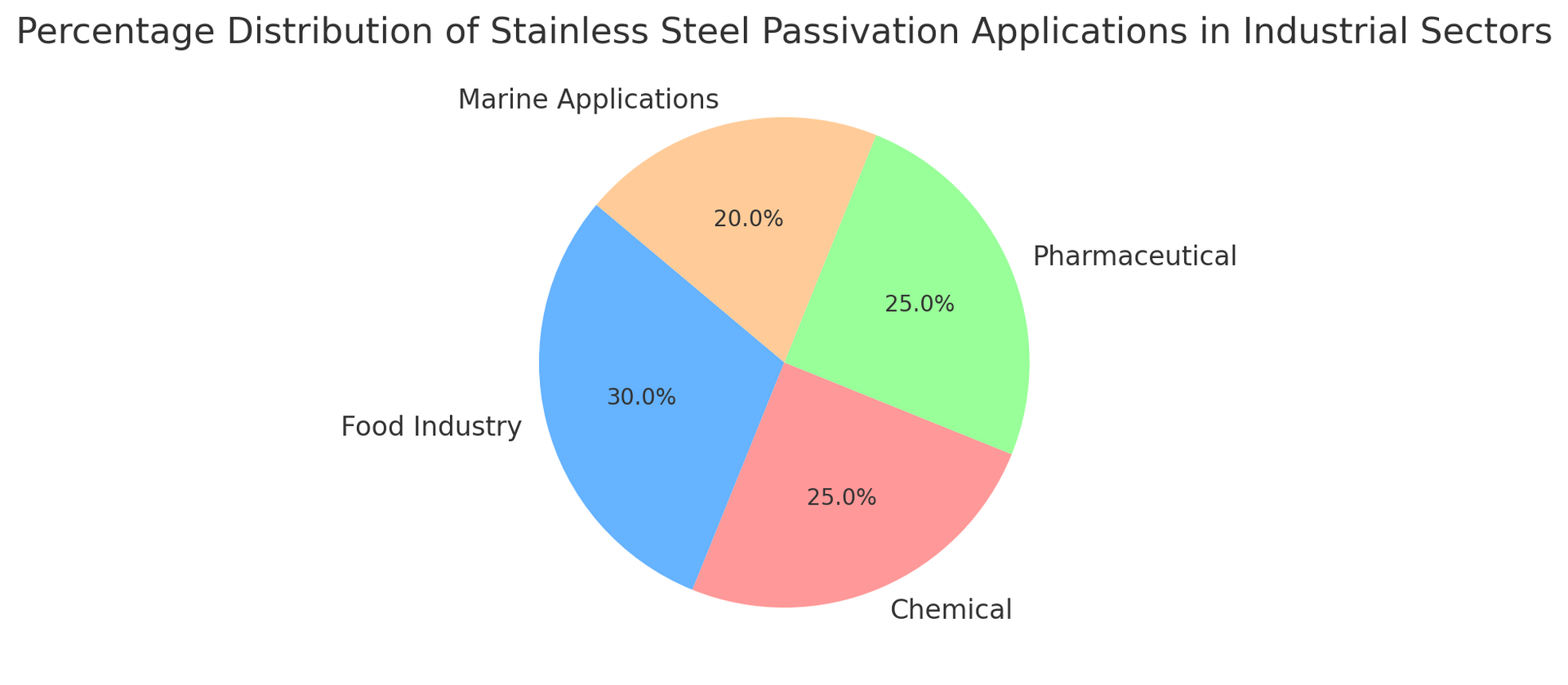
Chart: Distribution of Stainless Steel Passivation Applications
The chart below shows the percentage distribution of common stainless steel passivation applications across various industrial sectors:
This chart highlights how passivation is widely used in the food, chemical, pharmaceutical, and marine sectors, where corrosion resistance is essential to ensure the safety and durability of equipment.
Business Conclusion
Passivation is an essential treatment for stainless steel, maximizing its corrosion resistance and prolonging its service life, especially in aggressive environments.



